-
Hello guest! Are you an Apistogramma enthusiast? If so we invite you to join our community and see what it has to offer. Our site is specifically designed for you and it's a great place for Apisto enthusiasts to meet online. Once you join you'll be able to post messages, upload pictures of your fish and tanks and have a great time with other Apisto enthusiasts. Sign up today!
You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Hole in the head?
- Thread starter Spidy
- Start date
- Messages
- 2,768
- Location
- Wiltshire UK
Hi all,
Basically he has head butted something.
It could go either way, but he may well recover if you keep water quality up. I don't use melafix etc., but I add some <"Alder cones">, or Oak leaves, when this sort of thing happens.
cheers Darrel
It looks like a physical abrasion, rather than "Hole in the head".Is this hole in the head disease, or something else. Only fish affected.
Basically he has head butted something.
It could go either way, but he may well recover if you keep water quality up. I don't use melafix etc., but I add some <"Alder cones">, or Oak leaves, when this sort of thing happens.
cheers Darrel
Ade205
Active Member
- Messages
- 172
- Location
- Swadlincote, England
Just quick note from my experiences with hole in head.
I've been breeding German Rams for many years, I've literally reared thousands of them and as many of you I'm sure are aware, German Rams are extremely susceptible to HIH and other bacterial infection. By far the best solution I've found to rearing Ram fry and keeping adults also alive is UV sterilisation. I don't mean a canister or internal filter with a UV bulb, these past the water way to fast over the UV light, I'm talking about proper UV units that trickle the water over the light extremely slowly exposing algae, bacteria and pathogens to the UV for maximum possible time. Since using such units I have hardly any instances of Hole in head, or indeed Popeye which I've noticed tends to appear in conjunction with hih when you have a large number of fry or juvenile rams in grow out tanks. Before discovering this I would have a batch of fry every so often where bacterial infection would breakout and would be extremely hard to get on top off. Clean water is also vital, but I feel the bacteria once present multiplies to quick to control alone.
As to if the op's fish had hole in head or not, I don't think so but pretty certain bacterial infection was involved, maybe secondary to originally just wounding itself on decor.
Ade.
I've been breeding German Rams for many years, I've literally reared thousands of them and as many of you I'm sure are aware, German Rams are extremely susceptible to HIH and other bacterial infection. By far the best solution I've found to rearing Ram fry and keeping adults also alive is UV sterilisation. I don't mean a canister or internal filter with a UV bulb, these past the water way to fast over the UV light, I'm talking about proper UV units that trickle the water over the light extremely slowly exposing algae, bacteria and pathogens to the UV for maximum possible time. Since using such units I have hardly any instances of Hole in head, or indeed Popeye which I've noticed tends to appear in conjunction with hih when you have a large number of fry or juvenile rams in grow out tanks. Before discovering this I would have a batch of fry every so often where bacterial infection would breakout and would be extremely hard to get on top off. Clean water is also vital, but I feel the bacteria once present multiplies to quick to control alone.
As to if the op's fish had hole in head or not, I don't think so but pretty certain bacterial infection was involved, maybe secondary to originally just wounding itself on decor.
Ade.
- Messages
- 2,768
- Location
- Wiltshire UK
Hi all,
Do you know how old he was? Very few of my males have made it much past two years old, probably because I over-feed them and keep them fairly warm. A few of the females have made it to four.
cheers Darrel
Sorry to hear that.Hi Darrel.
Unfortunately it was something far more severe than that. His jaw was also funny and he eventually succumb. Lost another male same weekend. He jumped through the tiniest of gaps. :-(
Do you know how old he was? Very few of my males have made it much past two years old, probably because I over-feed them and keep them fairly warm. A few of the females have made it to four.
cheers Darrel
Ade, thank for the info and I agree about the things that GBRs are susceptible to. He did have a little white dot on his forehead before this came about so maybe you're right. An injury that got infected.
Darrel, not sure but if I had to guess, I'd say he was at least 18 months old. And I too keep them warm as they are in my Discus tank.
Out of curiosity, other than UV sterilization mentioned above, what do people use for bacterial infections? I know it's an open ended question dependent on the bacteria, but what do you guys use as a broad spectrum remedy?
Darrel, not sure but if I had to guess, I'd say he was at least 18 months old. And I too keep them warm as they are in my Discus tank.
Out of curiosity, other than UV sterilization mentioned above, what do people use for bacterial infections? I know it's an open ended question dependent on the bacteria, but what do you guys use as a broad spectrum remedy?
- Messages
- 315
- Location
- Finland
Hi,
I use various botanicals: leaves,seed pods and alder cones.
I have had one case of HITH in my tanks before adding these.
I use various botanicals: leaves,seed pods and alder cones.
I have had one case of HITH in my tanks before adding these.
- Messages
- 2,768
- Location
- Wiltshire UK
Hi all,
The real problem is with the pH scale, it looks like a simple linear scale, but it isn't really, it is both a ratio and a log^10 scale. Bases are "proton acceptors" and acids are "proton donors (H+)", and pH is the ratio of them, expressed as the negative log of the hydrogen ion (H+) activity.
Rather than just looking at pH I use another approach where you look at the magnitude of changes in water chemistry to assess whether something is likely to be harmful or not.
Hard water
In hard, carbonate buffered, water you have a lot of proton acceptors (bases), and these can combine with a lot of acids (proton donors) before pH changes. You need to add a lot of protons before the pH reduces, you need a large change in ionic composition. This is the situation in sea water or Lake Tanganyika, where small pH changes reflect large changes in water chemistry.
Soft Water
In soft water you don't have a reservoir of bases, and small additions of acids will cause rapid pH fall and small additions of bases will cause rapid pH rise. Small changes in water chemistry cause large changes in pH. This is the situation that will naturally happen for soft water fish during the diurnal cycle of variation in O2 and CO2.
This is from Rocha, RRA.; Thomaz, SM.*; Carvalho, P. & Gomes, LC. (2007) Modeling chlorophyll-α and dissolved oxygen concentration in tropical floodplain lakes (Paraná River, Brazil) Brazilian Journal of Biology 69:2

cheers Darrel
pH might go down a little bit, but you are always going to get pH fluctuation in soft water, pH is inherently unstable as you approach pure H2O.ButtNekkid, so you add all three at the same time? Doesn't it affect your pH wildly?
The real problem is with the pH scale, it looks like a simple linear scale, but it isn't really, it is both a ratio and a log^10 scale. Bases are "proton acceptors" and acids are "proton donors (H+)", and pH is the ratio of them, expressed as the negative log of the hydrogen ion (H+) activity.
Rather than just looking at pH I use another approach where you look at the magnitude of changes in water chemistry to assess whether something is likely to be harmful or not.
Hard water
In hard, carbonate buffered, water you have a lot of proton acceptors (bases), and these can combine with a lot of acids (proton donors) before pH changes. You need to add a lot of protons before the pH reduces, you need a large change in ionic composition. This is the situation in sea water or Lake Tanganyika, where small pH changes reflect large changes in water chemistry.
Soft Water
In soft water you don't have a reservoir of bases, and small additions of acids will cause rapid pH fall and small additions of bases will cause rapid pH rise. Small changes in water chemistry cause large changes in pH. This is the situation that will naturally happen for soft water fish during the diurnal cycle of variation in O2 and CO2.
This is from Rocha, RRA.; Thomaz, SM.*; Carvalho, P. & Gomes, LC. (2007) Modeling chlorophyll-α and dissolved oxygen concentration in tropical floodplain lakes (Paraná River, Brazil) Brazilian Journal of Biology 69:2

You should be able to get Magnolia grandiflora leaves, they sell these for Dart Frog Vivariums and there will be trees already in Australia (I know you have tight bio-security laws). Indian or Sea Almond (Terminalia catappa) would be another option, somewhere there is a thread about these trees growing widely along the coast in N. Queensland.Also, care to share the actual type of botanicals and where you purchase from so can can compare and find a supplier in Oz.
cheers Darrel
- Messages
- 315
- Location
- Finland
ButtNekkid, so you add all three at the same time? Doesn't it affect your pH wildly?
Also, care to share the actual type of botanicals and where you purchase from so can can compare and find a supplier in Oz.
Hi,
Seed pods come mainly from https://tanninaquatics.com/
https://tanninaquatics.com/collections/seed-pods/products/sterculia-pod
https://tanninaquatics.com/collections/seed-pods/products/cariniana-pod All of my apistos have laid eggs in these.
https://tanninaquatics.com/collections/seed-pods/products/nano-sterculia-pod
These pods last a LONG time so i mainly have to add leaves. Mostly Terminalia catappa from Ebay.
I´d prefer Magnolia leaves but they are hard to find cheaply.
And of course there are some driftwood and Apple tree branches sourced locally.
I don´t measure PH, only conductivity.
I am in the process of experimenting with deep leaf litter bed but that´s going take some time!
Thanks for the response guys.
Darrel, already using catappa leaves and see the effects on pH, hence the question.
ButtNekkid, you don't measure pH? I don't overly concern myself with it either in my fishroom as I only keep software fish, however, I tend to raise the pH again once it goes below 5.5 as I'm worried the beneficial bacteria will start to suffer potentially resulting in an Ammonia spike.
I know ammonia is less toxic in low pH (i.e. it's ammonium) but wouldn't a very low pH affect other parameters in the tank?
Darrel, already using catappa leaves and see the effects on pH, hence the question.
ButtNekkid, you don't measure pH? I don't overly concern myself with it either in my fishroom as I only keep software fish, however, I tend to raise the pH again once it goes below 5.5 as I'm worried the beneficial bacteria will start to suffer potentially resulting in an Ammonia spike.
I know ammonia is less toxic in low pH (i.e. it's ammonium) but wouldn't a very low pH affect other parameters in the tank?
- Messages
- 315
- Location
- Finland
Thanks for the response guys.
Darrel, already using catappa leaves and see the effects on pH, hence the question.
ButtNekkid, you don't measure pH? I don't overly concern myself with it either in my fishroom as I only keep software fish, however, I tend to raise the pH again once it goes below 5.5 as I'm worried the beneficial bacteria will start to suffer potentially resulting in an Ammonia spike.
I know ammonia is less toxic in low pH (i.e. it's ammonium) but wouldn't a very low pH affect other parameters in the tank?
Hi,
I have never measured PH. I´m lazy and if I get what I want, i.e. breeding apistos, I´m happy.
I bet Darrel will take over this thread
I stock my tanks with as much floating plants as is humanely possible. Those things love ammonia.
- Messages
- 2,768
- Location
- Wiltshire UK
Hi all,
I look after an analytical lab, with hundreds of thousands of £ worth of kit, but it is still really difficult to quantify what makes an aquarium successful. I've gone about this a different way now, which just consists of changing water on a regular basis, feeding the fish a good diet and having plenty of plants (including some with access to aerial gases) in some form of growth. Plants are the gift that keeps on giving.
The nitrification bit that @ButtNekkid alludes to is the recent research on nitrifying organisms which has found that they weren't what we thought they were and were much less limited by low pH and ammonia levels.
Basically since people were able to look for genes,
It has also been shown that low ammonia levels (<"an oligotrophic life style">) supports a much larger diversity of nitrifying organisms (if the link stops working the reference is Kits et al. "Kinetic analysis of a complete nitrifier reveals an oligotrophic lifestyle"), and it also looks likely that the complete nitrifier (COMAMMOX) Nitrospira is much more ubiquitous and important in nitrification than had been realised.
I think these should be available to everybody as well, Fowler et al. (2017) <"Comammox Nitrospira are abundant ammonia oxidizers in diverse groundwater‐fed rapid sand filter communities"> & Koch et al (2019) <"Complete nitrification: insights into the ecophysiology of comammox Nitrospira">.
cheers Darrel
Thanks for the response guys.......I don't overly concern myself with it either in my fishroom as I only keep software fish, however, I tend to raise the pH again once it goes below 5.5 as I'm worried the beneficial bacteria will start to suffer potentially resulting in an Ammonia spike.........I know ammonia is less toxic in low pH (i.e. it's ammonium) but wouldn't a very low pH affect other parameters in the tank?
I'm a lazy aquarist and a floating plant fan as well........I have never measured PH. I´m lazy and if I get what I want, i.e. breeding apistos, I´m happy.I bet Darrel will take over this threadwith his bacteria and archaeons! I stock my tanks with as much floating plants as is humanely possible. Those things love ammonia.
I look after an analytical lab, with hundreds of thousands of £ worth of kit, but it is still really difficult to quantify what makes an aquarium successful. I've gone about this a different way now, which just consists of changing water on a regular basis, feeding the fish a good diet and having plenty of plants (including some with access to aerial gases) in some form of growth. Plants are the gift that keeps on giving.
The nitrification bit that @ButtNekkid alludes to is the recent research on nitrifying organisms which has found that they weren't what we thought they were and were much less limited by low pH and ammonia levels.
Basically since people were able to look for genes,
- the range of organisms involved in nitrification, and the metabolic pathways utilised, have increased exponentially.
It has also been shown that low ammonia levels (<"an oligotrophic life style">) supports a much larger diversity of nitrifying organisms (if the link stops working the reference is Kits et al. "Kinetic analysis of a complete nitrifier reveals an oligotrophic lifestyle"), and it also looks likely that the complete nitrifier (COMAMMOX) Nitrospira is much more ubiquitous and important in nitrification than had been realised.
I think these should be available to everybody as well, Fowler et al. (2017) <"Comammox Nitrospira are abundant ammonia oxidizers in diverse groundwater‐fed rapid sand filter communities"> & Koch et al (2019) <"Complete nitrification: insights into the ecophysiology of comammox Nitrospira">.
cheers Darrel
The gist of what you're both saying (as I understand it) is that pH doesn't play such an important role because their are several groups of bacteria that are responsible for nitrification and if there is ever an ammonia spike you both use floating plants to combat it.
Firstly, I'd like to thank ButtNekkid for warning me about Darrel.
And secondly, I'd like to thank Darrel for ruining the rest of my week! There's a lot of reading in there.
There's a lot of reading in there.
Better get cracking.......
Firstly, I'd like to thank ButtNekkid for warning me about Darrel.
And secondly, I'd like to thank Darrel for ruining the rest of my week!
Better get cracking.......
My head almost exploded reading those (not in full admittedly). Thanks Darrel.
I also found this study by Thi Thu Huyen Le et. al. (2019)<Kinetics and simulation of nitrification at various pH values of a polluted river in the tropics>useful to read and is more closely related to the effects of pH.
I also found this study by Thi Thu Huyen Le et. al. (2019)<Kinetics and simulation of nitrification at various pH values of a polluted river in the tropics>useful to read and is more closely related to the effects of pH.
- Messages
- 2,768
- Location
- Wiltshire UK
Hi all,
Before we had DNA they were thought to be bacteria.
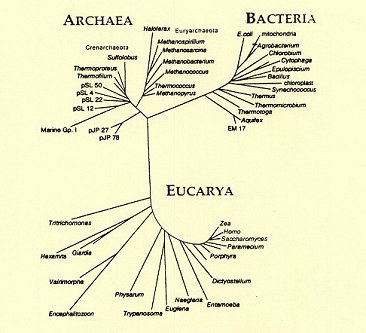
There is also "Lehtovirta-Morley, L. <"Ammonia oxidation: Ecology, physiology, biochemistry and why they must all come together">, FEMS Microbiology Letters, Volume 365, Issue 9, May 2018".
It is well worth a read.
cheers Darrel
Yes, pretty much. We now know that a one of the three fundamental divisions of life are the Archaea, and that they are a lot more common than we realised, and a lot more important in nitrification. Have a look at <"Bacteria revealed">.The gist of what you're both saying (as I understand it) is that pH doesn't play such an important role because their are several groups of bacteria that are responsible for nitrification and if there is ever an ammonia spike you both use floating plants to combat it.
Before we had DNA they were thought to be bacteria.

I'm just a floating plant obsessive. It is only a matter of time before I start knocking on random doors and asking people if they've already heard the "good news" about Frogbit, but I'm a botanist, not a real scientist, I have to ask @regani about the difficult questions, he is a chemist.Firstly, I'd like to thank ButtNekkid for warning me about Darrel.
Yes, some of them are a bit impenetrable, the <"Bagchi paper"> is probably the best to start with.And secondly, I'd like to thank Darrel for ruining the rest of my week!There's a lot of reading in there.
Better get cracking.......
There is also "Lehtovirta-Morley, L. <"Ammonia oxidation: Ecology, physiology, biochemistry and why they must all come together">, FEMS Microbiology Letters, Volume 365, Issue 9, May 2018".
It is well worth a read.
That is an interesting paper, I'm pretty sure they are right about enhanced nitrification rates at higher pH levels, but it isn't particularly relevant to us. The river is so polluted it is more similar to the effluent from a sewage works than our to tanks.I also found this study by Thi Thu Huyen Le et. al. (2019)<Kinetics and simulation of nitrification at various pH values of a polluted river in the tropics>useful to read and is more closely related to the effects of pH.
cheers Darrel
Similar threads
- Replies
- 4
- Views
- 409

